Le Dr Phatcharasak a réalisé plus de 20 000 interventions et possède 33 ans d'expertise en thérapie cellulaire. Le traitement peut coûter environ 1 800 $ pour 5 millions de CSM de cordon ombilical, incluant généralement les examens préopératoires, les honoraires médicaux et les transferts. Ce spécialiste certifié en chirurgie plastique a été formé à l'Université de Khon Kaen. L'ID Clinic détient l'accréditation du Royal College of Surgeons de Thaïlande.
Comment ça fonctionne
Quel est le Coût pour votre Thérapie de rajeunissement des cellules souches en Thaïlande ?
Avantages clés
- Profitez de la thérapie de rajeunissement par cellules souches en Thaïlande à un coût inférieur de 40-60% par rapport à la France.
- Options disponibles: cellules souches mésenchymateuses, cellules souches embryonnaires, et cellules souches adultes.
- Évaluation gratuite par un médecin expert pour déterminer la meilleure option de rajeunissement cellulaire pour vous.
- 100% garantie de protection des données et de confidentialité.
- Accès à des offres exclusives de cliniques renommées en Thaïlande de $2,191.
| Thaïlande | Turquie | Pologne | |
| Thérapie de rajeunissement des cellules souches | de $3,500 / 129,500฿ | de $1,297 / 47,989฿ | de $5,500 / 203,500฿ |
| Thérapie par exosomes | de $2,000 / 74,000฿ | de $2,200 / 81,400฿ | de $2,100 / 77,700฿ |
| Rajeunissement avec ses propres cellules souches | de $6,000 / 222,000฿ | de $6,000 / 222,000฿ | de $5,000 / 185,000฿ |
| Rajeunissement avec des cellules souches de donneurs | de $5,500 / 203,500฿ | de $3,000 / 111,000฿ | de $5,000 / 185,000฿ |
| Mésothérapie | de $350 / 12,950฿ | de $300 / 11,100฿ | de $250 / 9,250฿ |
Ne Manquez pas les Offres Exclusives de thérapie de rajeunissement des cellules souches en Thaïlande en avril 2026
Le Dr Wanna Supasirilak apporte 12 ans d'expertise dans les traitements par cellules souches, ayant réalisé plus de 900 interventions. Le forfait 5MC peut coûter entre 75 000 et 89 000 THB, incluant généralement la thérapie cellulaire, le PRP, l'anesthésie et les soins postopératoires. L'hôpital Wansiri est reconnu par le Royal College of Surgeons de Thaïlande et accueille 2 000 patients par an. Des systèmes de filtration d'air avancés et des blocs opératoires stériles garantissent la sécurité pendant le séjour hospitalier d'une journée.

Le Dr Chollawat Thongthaisin apporte 19 ans d'expertise en tant que spécialiste de la médecine régénérative certifié par l'American Board, avec plus de 900 interventions réalisées. La thérapie de rajeunissement par cellules souches à la Vega Clinic peut coûter environ 8 500 à 12 000 USD, couvrant généralement la consultation, les examens, la thérapie, les perfusions de vitamines, l'oxygénothérapie et les transferts VIP. La clinique accueille chaque année plus de 200 patients internationaux grâce à une approche multidisciplinaire utilisant des cellules souches autologues pour des traitements anti-âge personnalisés.

Le Dr Sarunya Yuthagovit a réalisé plus de 900 traitements par cellules souches sur 11 ans, se spécialisant dans les thérapies régénératives. Le forfait de rajeunissement de la clinique coûte environ 5 200 $, couvrant généralement l'intervention, les analyses de laboratoire et le parking. Le Dr Sarunya dirige les programmes de neurologie et de cellules souches en mettant l'accent sur la sécurité clinique. EDNA Wellness maintient un taux de recommandation des patients de 98 % pour ses traitements à haute concentration cellulaire.
- Karin Kulwacharamongkon poursuit un doctorat en médecine anti-âge et régénérative avec une formation esthétique avancée.
- Harmony Life Center propose une technologie de scan facial en 3D dans le cadre de sa thérapie de rajeunissement par les cellules souches du visage.
- Services inclus : zone café, parking, scan facial 3D, consultation avec un dermatologue.
- Informations sur le séjour : Aucun hébergement ou séjour à l'hôtel n'est inclus dans le prix.
Découvrez les Meilleures Cliniques pour Votre Thérapie de rajeunissement des cellules souches en Thaïlande : 8 Options Vérifiées et Prix
Clinique Vital Glow Skin & Aesthetic à l’Hôpital Navamin 9
Ariyapon Lortaraprasert
Clinique de chirurgie plastique Lelux
Dr. Chen Surgery Hospital International Center
Chen Chaicharncheep
Aperçu de thérapie de rajeunissement des cellules souches en Thaïlande
- Thérapie de rajeunissement des cellules souches - de $3,500Rajeunissement avec des cellules souches pour améliorer la santé globale et réduire les affections liées à l'âge.
Jour 1
- Arrivée à l'aéroport.
- Transfert vers un hôtel 4*.
Jour 2
- Visite à la clinique pour une première consultation.
- Rencontre avec le spécialiste des cellules souches.
- Subir les tests requis pour l’adéquation du traitement.
Jour 3
- Discussion des résultats des tests avec le spécialiste.
- Préparation à la thérapie de rajeunissement des cellules souches.
Jour 4
- Suivre une thérapie de rajeunissement par cellules souches.
- Le processus prend environ 2 à 3 heures.
- L'anesthésie locale est généralement utilisée.
Jour 5
- Repos et récupération à l'hôtel.
- Assistance médicale disponible 24h/24 et 7j/7, si nécessaire.
Jour 6
- Consultation post-thérapeutique avec le spécialiste.
- Discussion sur les soins post-thérapeutiques et la réadaptation.
Semaine 1 - Semaine 4
- Les activités physiques légères sont encouragées.
- Évitez les activités ou les sports intenses.
- Suivis réguliers avec le spécialiste.
À partir de la semaine 5
- Retour progressif aux activités et sports réguliers.
- Continuez les suivis réguliers avec le spécialiste.
- Résultats complets visibles après 2-3 mois.
Veuillez noter que la situation et le rétablissement de chaque patient peuvent varier. Consultez toujours votre spécialiste pour obtenir des conseils personnalisés.
- Dépôt Bookimed: $200 ou €200.
- Dépôt de la clinique: payez un pourcentage fixe à l'avance.
Obtenez une Évaluation Médicale pour Votre Thérapie de rajeunissement des cellules souches en Thaïlande : Consultez Maintenant 31 Médecins Expérimentés
Voir tous les médecinsDr Phatcharasak Kraisornphongsakul Md
Le Dr Phatcharasak Kraisornphongsakul Md est spécialisé en thérapie de rajeunissement par cellules souches à la Clinique ID de Bangkok, se concentrant sur les traitements anti-âge.
- Vaste expérience en médecine régénérative
- Expert en thérapies par cellules souches de pointe
- Engagement envers des plans de traitement personnalisés
Chollawat Thongthaisin
Le Dr Chollawat est spécialisé en médecine régénérative et en rajeunissement par cellules souches à la Vega Clinic – soutenue par BDMS, le plus grand groupe hospitalier privé de Thaïlande.
- Certifié en médecine régénérative par l'American Board of Regenerative Medicine
- Médecin chef chez Vega Stem Cell avec des recherches en cours dans le domaine
- Formé en radiologie à l'hôpital Thammasat – améliorant la précision du diagnostic
- Travaille dans les départements anti-âge et de chirurgie plastique de l'hôpital Jomtien
Napat Jirattitikarntavorn
Le Dr Napat est spécialisé dans les solutions anti-âge personnalisées à la Menness Wellness Clinic, combinant la médecine esthétique et les thérapies régénératives.
- Certifié en médecine esthétique et en traitements anti-âge
- Expert en thérapie PRP et IV (intraveineuse) pour le rajeunissement
- Se concentre sur des approches holistiques, y compris l'équilibre hormonal
- Fournit une planification anti-âge personnalisée pour chaque patient
Doctor Sirintip Jira-adisai
Le Dr Sirintip Jira-adisai est une spécialiste renommée en médecine anti-âge, en dermatologie et en médecine régénérative, aidant les patients à obtenir une peau jeune et saine.
- Spécialisée dans la thérapie de rajeunissement par cellules souches pour l'anti-âge
- Exerce à l'Asia Cosmetic Hospital à Bangkok
- S'engage à se tenir au courant des dernières avancées
- Reconnue pour son expertise en médecine anti-âge et en dermatologie
Histoires vidéo des patients de Bookimed
Avis sur Bookimed : Découvrez l'Expérience des Patients
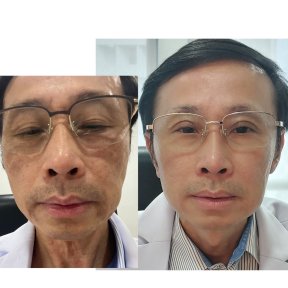
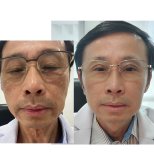
Tous les avisThérapie de rajeunissement des cellules souches: Photos avant et après
Voir tous les Avant & AprèsPartager ce contenu
FAQ de thérapie de rajeunissement des cellules souches en Thaïlande
Is stem cell therapy for anti-aging legal and regulated in Thailand?
Stem cell therapy for anti-aging is legal and strictly regulated in Thailand under the Thai Food and Drug Administration and the Medical Council of Thailand. Procedures must occur in licensed facilities using cells processed in Good Manufacturing Practice (GMP) certified laboratories by board-certified regenerative medicine physicians.
- Regulatory oversight: The Ministry of Public Health monitors safety, quality, and ethical standards for treatments.
- Clinical standards: Clinics like Asia Cosmetic Hospital hold Joint Commission International (JCI) accreditation for safety.
- Cell sourcing: Protocols typically use autologous cells from patient fat or certified umbilical cord mesenchymal cells.
- Physician qualifications: Many Thai specialists, including those at Vega Clinic, hold American Board of Regenerative Medicine certification.
Bookimed Expert Insight: Thailand’s regulatory framework uniquely permits expanded cell cultures, allowing for high-dose treatments of 40 to 60 million mesenchymal stem cells. Clinics like Vega Clinic or EDNA Wellness offer these systemic protocols which are often restricted in Western jurisdictions. This high-volume experience is reflected in the market data, where Thailand ranks second globally for requested procedures on our platform.
Patient Consensus: Patients emphasize the importance of verifying Medical Council registration and starting with video consultations to confirm lab-tested cell viability. While clinics advertise freely under cosmetic exemptions, experienced travelers recommend securing medical insurance that covers regenerative treatments for added peace of mind.
What types of stem cells are used?
Anti-aging clinics in Thailand primarily utilize umbilical cord-derived mesenchymal stem cells (UC-MSCs) and Wharton's Jelly-derived MSCs. These allogeneic donor cells are preferred for their high proliferative capacity and potency. Leading facilities often combine these treatments with intravenous NAD+ therapy or hyperbaric oxygen to enhance regenerative outcomes.
- Cell source: Most providers use donor-sourced umbilical cord mesenchymal stem cells (UC-MSCs).
- Cell potency: Multipotent MSCs can differentiate into various tissues like bone, cartilage, and fat.
- Adjunct therapies: Treatments often include NAD+ infusions, vitamin drips, or hyperbaric oxygen sessions.
- Common dosages: Packages typically offer protocols ranging from 25 million to 60 million MSCs.
Bookimed Expert Insight: While many global clinics still rely on harvesting a patient's own fat or bone marrow, Thai centers like Vega Clinic and EDNA Wellness have shifted almost entirely to off-the-shelf UC-MSCs. This transition allows doctors to bypass invasive extraction surgeries and provide standardized, high-volume cell counts that are often difficult to achieve with autologous (own) cells in older patients.
Patient Consensus: Patients emphasize the importance of requesting a Certificate of Analysis to verify cell viability and sterility. Many report a significant energy boost lasting 3 to 6 months after receiving high-dose MSC infusions.
How is the treatment administered and is there any downtime?
Anti-aging stem cell therapy in Thailand occurs via intravenous infusion or localized injections using umbilical cord mesenchymal cells. Sessions typically last 1 to 3 hours over 3 to 5 days. Patients experience minimal downtime of 1 to 3 days, returning to normal activities within 72 hours.
- Administration methods: Specialist doctors perform IV infusions, local facial injections, or use mesotherapy guns.
- Treatment duration: Procedures involve 1 to 3 hour sessions scheduled over 3 to 5 days.
- Recovery timeline: Most patients resume work in 48 hours but may feel fatigue for 3 days.
- Side effects: Mild flu-like detox symptoms or Herxheimer reactions can last up to 7 days.
Bookimed Expert Insight: High-end Bangkok clinics like Vega Clinic often combine MSCs with hyperbaric oxygen or NAD+ drips. These additions aim to shorten the typical 3-day fatigue window. Choosing packages with these recovery boosters allows some patients to return to light gym activity within 24 hours.
Patient Consensus: Many travelers report feeling ready for light sightseeing within 2 days. Some experience deeper fatigue after lower-cost treatments, so planning a 7-day Thailand stay ensures proper monitoring and hydration.
How long do the results last?
Stem cell rejuvenation results in Thailand typically last 6 to 18 months depending on the cell count and delivery method. Patients often see peak improvements between 3 and 6 months. Using 25 to 60 million mesenchymal stem cells helps maintain cellular repair and systemic anti-aging effects.
- Cell quantity: Higher doses like 60 million MSCs typically provide more sustained systemic rejuvenation.
- Co-therapy impact: Adding NAD+ drips or hyperbaric oxygen therapy can extend results beyond 18 months.
- Technique used: Intravenous infusions offer systemic benefits while 3D-guided facial injections target localized skin quality.
- Maintenance schedule: Most patients schedule top-up treatments every 9 to 12 months to maintain vitality.
Bookimed Expert Insight: Data from top Bangkok clinics shows a clear link between cell count and longevity. Packages with 60 million MSCs at Vega Clinic often yield more durable results than 10 million cell facial boosters. For the best value, look for bundles including hyperbaric oxygen, as this specialized aftercare supports cell survival and enhances the overall duration of the treatment.
Patient Consensus: Many patients find that results are highly individual and strongly influenced by daily habits. While the initial glow is visible early on, maintaining a healthy diet and consistent sleep schedule is reported to nearly double the observed longevity of the rejuvenation effects.